ALTALAB is an engineering consultancy company located in Milan, Italy. We are active in various fields of engineering...
What's our story
A short description of Altalab's history of creation
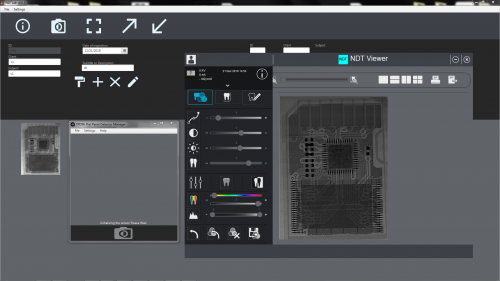
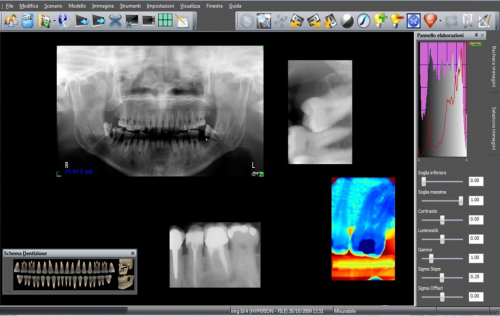
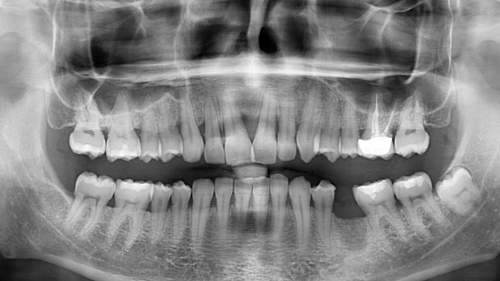
ALTALab is a contract R&D services company, founded in 2007 initially by 5 engineers with a specific background in the development of X-ray digital systems for dental and medical radiology, in Milan, ITALY.

ALTALAB leverages on a qualified network of external professionals and institutions and is committed to delivering to its customers high tech and quality solutions for products and services.
Among the strengths is the versatility and creativity of the team, who has gained his diversified experience cooperating with various multinational as well as small/medium industries.
ALTALAB pursues the maximum customer satisfaction as well as well as the flexibility, opening and scalability of the proposed technology platforms.
ALTALab is a contract R&D services company, founded in 2007 initially by 5 engineers with a specific background in the development of X-ray digital systems for dental and medical radiology, in Milan, ITALY.

ALTALAB leverages on a qualified network of external professionals and institutions and is committed to delivering to its customers high tech and quality solutions for products and services.
Among the strengths is the versatility and creativity of the team, who has gained his diversified experience cooperating with various multinational as well as small/medium industries.
ALTALAB pursues the maximum customer satisfaction as well as well as the flexibility, opening and scalability of the proposed technology platforms.
What do we do in
ALTALAB
A general description of the scope of activities of Altalab
Here our Mission is Creating added value for our customers by design of innovative products and services and “lean” development and manufacturing processes, which combine technology and expertise in building solutions characterized by effectiveness and quality.
In ALTALAB we are focused on Establishing privileged and exclusive relations with our
customers in the particular market sector, aiming to achieve improvement and added value in the following areas :
- Product Portfolio, definition and expansion
- Intellectual Property, strategy and planning
- Product Development Process, innovation and time to market
- Product Quality, profitability and customer satisfaction
- Manufacturing Process, lean tools and flexibility
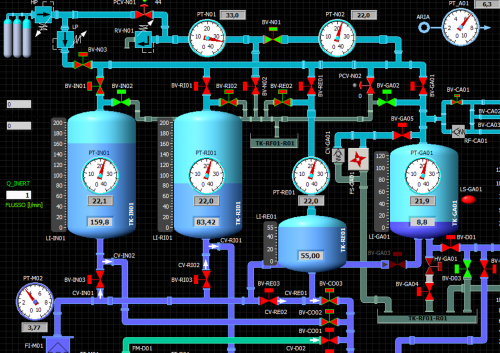
ALTA Lab provides to its customers integrated services
along the whole product development cycle, from the
ideation of the new equipment to the design of efficient
the manufacturing process at the customer’s site, among which:
- Product design, development, prototyping
in accordance with the customer’s product strategic planning
- Product Certification
against relevant international standards, in particular for medical
devices in the European market (CE) and North American market
(FDA, UL and CSA)
- Design, implementation and validation of the manufacturing
process, in accordance with the lean production principles